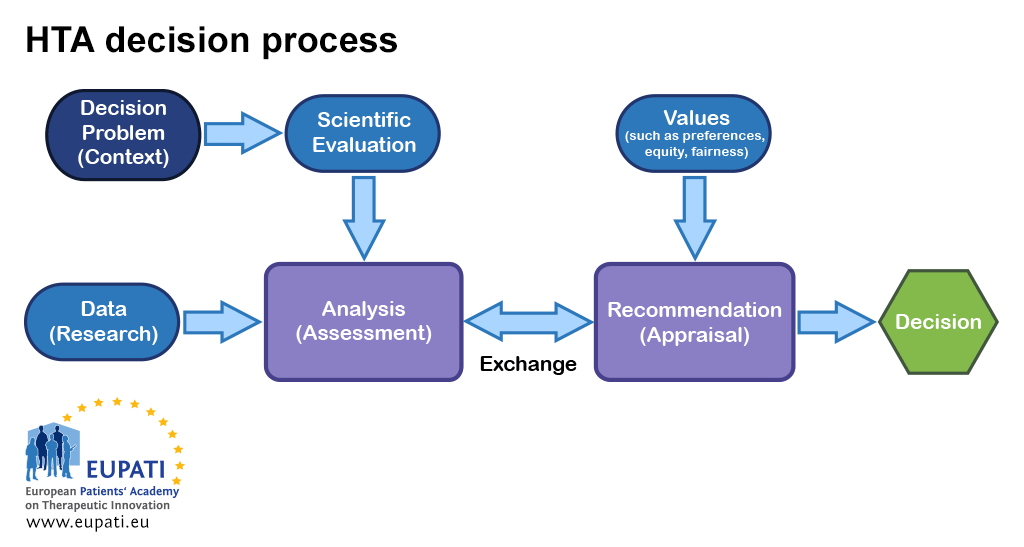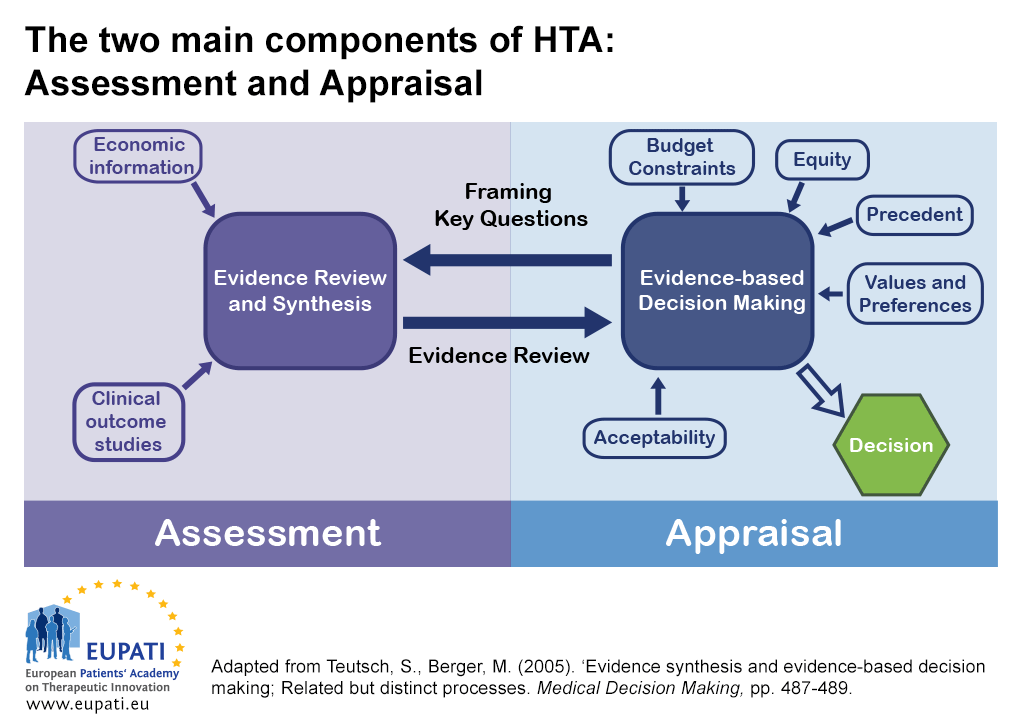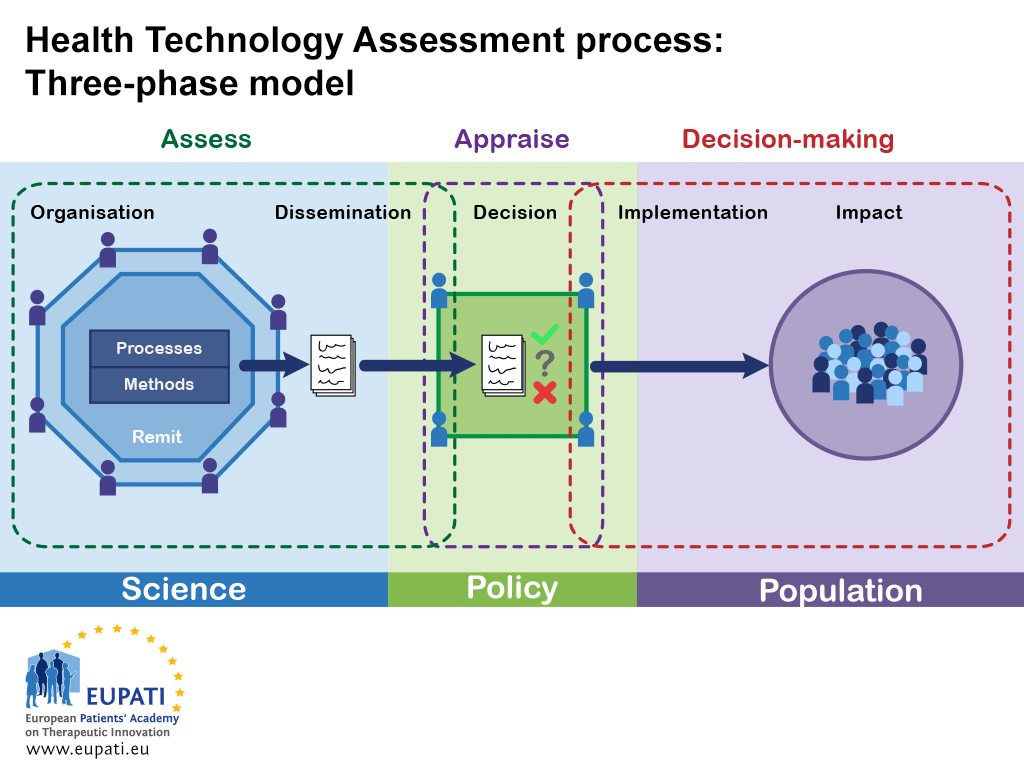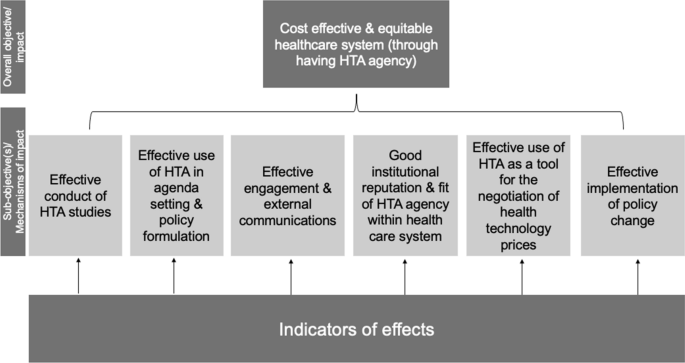
Assessing the performance of health technology assessment (HTA) agencies: developing a multi-country, multi-stakeholder, and multi-dimensional framework to explore mechanisms of impact | Cost Effectiveness and Resource Allocation | Full Text

Differences in Evidentiary Requirements Between European Medicines Agency and European Health Technology Assessment of Oncology Drugs—Can Alignment Be Enhanced? - Value in Health

Establishing a national HTA program for medical devices in Italy: Overhauling a fragmented system to ensure value and equal access to new medical technologies - ScienceDirect

Similarities and Differences in Health Technology Assessment Systems and Implications for Coverage Decisions: Evidence from 32 Countries | PharmacoEconomics - Open

Correspondence analysis biplot illustrating the relative associations... | Download Scientific Diagram
Concordance on ATV assessments among HTA bodies (Note: the size of the... | Download Scientific Diagram

Shaping a research agenda to ensure a successful European health technology assessment: insights generated during the inaugural convention of the European Access Academy | Health Economics Review | Full Text

Establishing a national HTA program for medical devices in Italy: Overhauling a fragmented system to ensure value and equal access to new medical technologies - ScienceDirect